More than 200 individuals are on waiting lists for the $373,000 treatment as the clinic struggles to resolve issues with the insurance agency.
According to a report distributed last Thursday (Dec. 14) in Bloomberg, Gilead Sciences’ blood disease treatment was given to only five patients within two months of its approval. Firm enrollments for Yescart – a CAR T-cell therapy for giant B-cell lymphomas – reportedly exceeded 200 individuals, with some exceptionally debilitated patients passing before they could receive the treatment.
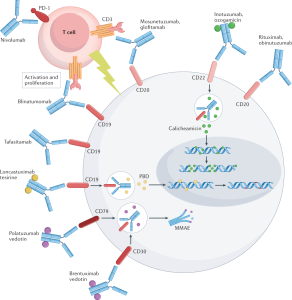
Yescarta, originally created by Kite Pharma — a California startup that recently bought Gilead for nearly $12 billion in real money — is only the second CAR T-cell treatment to be approved by U.S. specialists at 15 emergency clinics approved to run the new treatment, asking for installment approval of $373,000 for treatment from insurance agencies, including Medicare and Medicaid. “There are no charge codes for this,” Michael Bishop, head of the cellular therapy program at the University of Chicago Medicine Hospital, told Bloomberg. “It was extreme, honestly.
See “FDA Approves Second Car T-Cell Therapy”.
While some medical clinics are delaying getting therapy until protection is in place, others are exploring options. Specialists at the MD Anderson Cancer Center — which has more than 100 on the list — allow patients to sign a waiver that says the actual patients will take care of everything for the therapy in case the guarantors deny it. Stanford University, meanwhile, recognizes patients regardless of their protection status. “My organization is taking on the challenge because it’s the right thing to do,” Stanford physician David Miklos told Bloomberg. It’s a colossal gamble, though, and it keeps me up late at night.
At the time of its approval in October, Yescarta was hailed as “a breakthrough in advancing a whole new logical worldview for the treatment of serious diseases,” in an explanation by US Food and Drug Administration Commissioner Scott Gottlieb. David Maloney, clinical leader of cellular immunotherapy at the Fred Hutchinson Cancer Research Center, predicted on NBC that “a lot of lives will probably be saved in this sense in the next few years.”
See “Vehicle T-Cell Race”.