The new tool, called PRISM, draws on virus-host interactions and the DNA repair pathway to help researchers visualize how cellular stress can contribute to neurodegenerative disease.

Unlike most cells, mature neurons do not divide. Because mutations often arise through error-prone mechanisms of cell division, scientists once believed that these neurons could not accumulate many mutations. Recently, however, scientists have discovered that genotoxic stress—which causes DNA damage—can induce somatic mutations in brain cells, and are now trying to link this phenomenon to neurodegenerative diseases.
One such neuroscientist, Xiao-Hong Lu of Louisiana State University Health Shreveport, found that many existing tools for probing DNA damage depend on cellular replication pathways that are not present in neurons. “We can’t directly use these methods,” he says, so he created his own, “a sensor specifically for the brain.”
Lu’s genetic sensor, called PRISM (Viral Proxy Probe for DNA Instability Surveillance/Repair in Somatic Brain Mosaic), uses a viral vector to mark cells that have initiated their DNA damage response (DDR), a set of signaling cascades that regulate repair DNA and cell cycle checkpoints following genotoxic stress. In many cells, the DDR not only recognizes DNA double-strand breaks, a common form of DNA damage; it also acts as a form of innate immunity, perhaps because viruses break DNA to integrate into host genomes.
The PRISM viral vector enters many neurons, but replicates more readily in cells whose DDR is already activated and therefore not available to confront the virus. The PRISM genome is engineered to include a repetitive sequence with a frameshift mutation. Once inside the cell, the host’s DNA replication machinery reverses the frameshift while converting single-stranded DNA to double-stranded, triggering the production of a fluorescent protein that allows researchers to monitor PRISM-infected neurons in vivo.
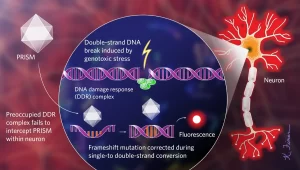
To label neurons that have undergone genotoxic stress, the researchers developed an in vivo sensor using an adeno-associated viral vector, called PRISM. Because the cellular DNA damage response (DDR), which is activated in response to stressors such as environmental toxins or the accumulation of misfolded proteins, also responds to invading pathogens, PRISM has an easier time transfecting cells whose response mechanisms on damage are biased by existing DNA damage. . Once inside, the virus hijacks the neuron’s DNA replication machinery, which reverts the artificial shift mutation in the virus, thereby triggering the production of a fluorescent protein that can be observed under a microscope. WEB | PDF
© KELLY FINANCIAL
The researchers tested PRISM in mice exposed to paraquat, a herbicide associated with an increased risk of Parkinson’s disease (PD), and in a humanized mouse model of a non-hereditary form of PD characterized by aggregation of the misfolded protein α-synuclein.
The resulting fluorescence reliably marked cells that had activated their DDR pathways, and these cells continued to fluoresce even after paraquat was removed or α-synuclein levels were reduced. In both experimental mouse populations, PRISM signaled damage in the most affected cell type of PD, dopaminergic neurons in the substantia nigra. Lu analyzed the postmortem brains of six sporadic PD patients and found that up to 40 percent of nigral dopaminergic neurons had double-strand breaks—a result he calls “quite prominent.”
Diane Shao, a physician and scientist at Harvard Medical School and Boston Children’s Hospital who was not involved in the study, notes that PRISM is an error-prone but “elegant” tool and says that “overall, the system is very simple” to use. . But he adds that the marker only marks DNA double-strand breaks, which “is only one type in the scheme of DNA damage.”
Lu tells The Scientist that PRISM has uses beyond disease research. The team recently launched a NASA-funded project to investigate what happens to mice exposed to simulated cosmic radiation, for example. “We know that cells have damaged DNA,” he says.